Screening (about 6 weeks)
The trial staff will do some tests and checks to see if the trial is a good fit for you.
The APPROACH trial is exploring an investigational psychedelic medication, combined with psychological support, to learn how effective it might be at reducing symptoms of major depressive disorder (MDD).
In recent years there has been growing interest in the potential role psychedelic medications could play in the treatment of mental health conditions.
The investigational study medicine, called CYB003, is similar to psilocin (the active ingredient in Psilocybe mushrooms). Psychedelic medications, at certain doses, may cause an altered state of consciousness, unique thoughts, and changes in your senses, such as hearing, seeing, tasting, and/or smelling things that are not real. Psychological support and CYB003 given together may help people reach deeper into their thoughts, promote healing, and may be effective in treating MDD.
A licensed therapist with specialized training in EMBARK, a psychological support model for MDD, will work with participants during several psychological support sessions before and after the dosing sessions. They, along with a second session monitor, will also attend the dosing sessions for both psychological support and safety purposes.
All medicines must be approved by the appropriate regulatory authorities in any country or region before they can be used there. CYB003 is not yet approved in any country.
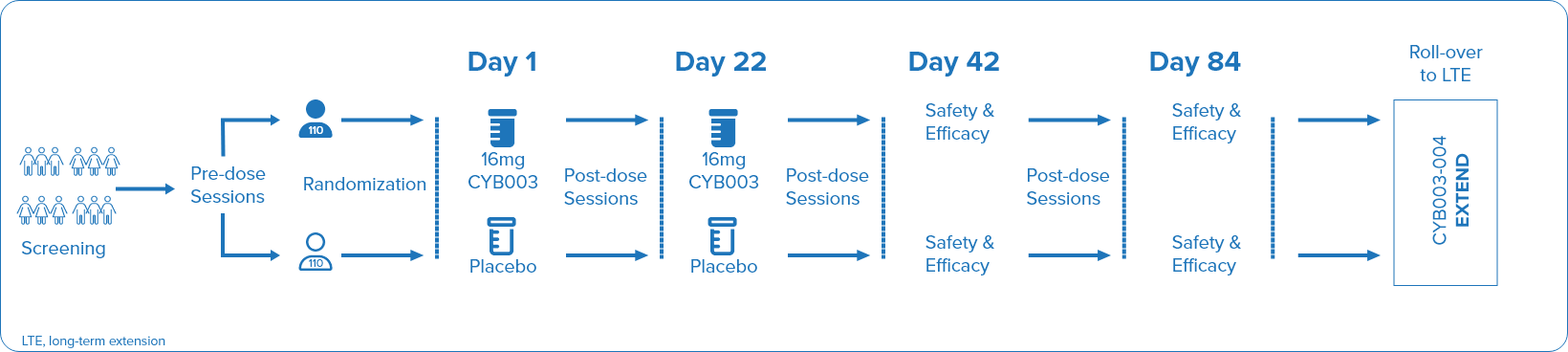
CYB003 is given as a capsule to be swallowed whole. Participants will take the investigational study medicine at dosing sessions during clinic visits. They will have two dosing sessions in total, about three weeks apart.
During the dosing sessions, you may experience changes in how you hear and see things, feel, and think for several hours. Both of your session monitors will stay with you to provide support and take any necessary safety measures. You will be accompanied home by a trusted adult (friend or family member).
Participation is expected to last about four and a half months.
You will have a total of:
Most of the visits in this trial will last at least two hours. The two visits at which you have dosing sessions are expected to last at least 8 hours.
You will be assigned randomly (like flipping a coin) to receive either CYB003 or a placebo combined with psychological support. A placebo is an inactive treatment which looks the same as the investigational study medicine but does not contain any active ingredient.
You will have a 50% chance of receiving the investigational study medicine and a 50% chance of receiving a placebo. Neither you nor the trial doctors will know which treatment you are taking, this is known as a ‘double-blinded’ trial.
While we understand that using placebos and blinding can cause some uncertainty, they are vital to confirm that any effects seen in the trial are due to the investigational study medicine.
You will also continue to take any standard antidepressant medication you are already taking.
Double-blinded controlled trials are considered the gold standard of evidence in medical research.
The trial staff will do some tests and checks to see if the trial is a good fit for you.
You will receive three psychological support sessions to prepare for your first dose.
The two dosing sessions will last about 8 hours, during which time session monitors will provide psychological support and ensure your safety.
After each dose, during a 3-week period you will have:
You will have two clinic visits and two remote visits to monitor your wellbeing and MDD symptoms.
Participants will also have the option to join a long-term extension to the trial if they meet the requirements, where all participants can receive CYB003 with psychological support once they meet the requirements for dosing – there is no placebo group in this extension trial. The extension will last about 43 weeks.
During the clinical trial, a team of doctors, researchers and healthcare professionals will take care of you and check on your progress. What happens at each clinic visit will vary, but activities may include:
A review of your health including any ongoing conditions you may have or medications you are currently using.
Discussions about your symptoms and recent depressive episodes.
Swallowing a capsule of either CYB003 or placebo.
An overall check of your body’s condition, including general appearance and tests of various systems.
A check of your blood pressure, pulse rate, respiratory rate, and temperature.
Samples of your blood and urine will be collected for health tests.
A test of your heart’s electrical activity and function using wired electrodes attached to your skin.
You will answer a series of questions about your mental health, symptoms, anxiety, and quality of life.
will include: